
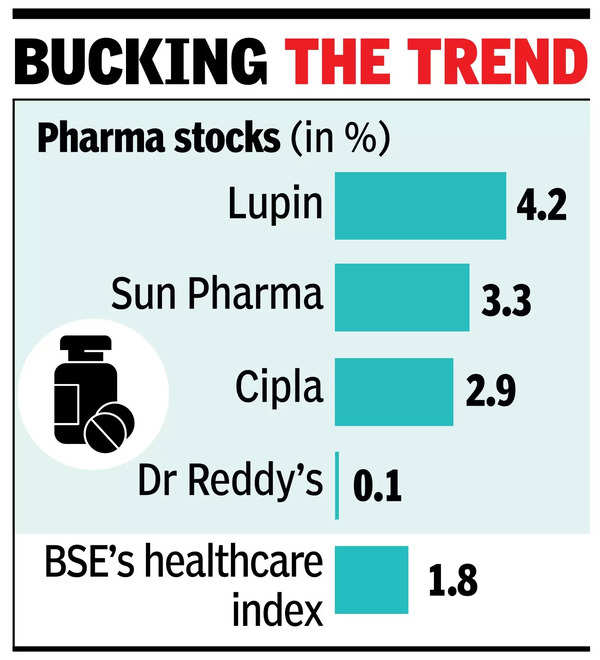
NEW DELHI: The Trump administration’s decision to exempt tariffs on pharma imports from India calmed industry nerves, delivering relief to a sector tied to India’s $9 billion-plus US export market. This move underscores the critical role of India’s domestic pharma industry in supplying cost-effective, life-saving generic medicines, with the US relying on India for nearly 30% of its prescription medications. The sector was jittery with the expectation of a 10% tariff on Indian pharma exports to the US. However, some analysts caution that this relief may be short-lived, saying tariffs are not completely ruled out for the future.
Sudarshan Jain, secretary general, Indian Pharmaceutical Alliance, says, “India and the US share a strong and growing bilateral trade relationship, with a shared vision to double trade to $500 billion under the Mission 500 initiative. Pharmaceuticals remain a cornerstone of this partnership, as India plays a vital role in global and US healthcare by ensuring a steady supply of affordable medicines.”
Meanwhile, the medical device sector wasn’t as fortunate, facing the brunt of new tariff challenges and cost pressures. “The imposition of a 26% reciprocal tariff on Indian medical device exports to the US may pose a significant challenge to the sector’s growth. Historically, India has been a key supplier of cost-effective, high-quality medical devices to the US, primarily in low-value, high-volume consumables categories. However, this new tariff may possibly impact domestic medical devices exports, and we have to explore windows of opportunities where the US has been seeking to diversify its supply chain dependence on any one nation,” Rajiv Nath, forum coordinator, AiMeD, said.
In 2023-24, India’s medical device exports to the US stood at around $714 million, while US exports to India were double, around $1,500 million, as per data shared by the Exports Promotion Council of Medical Devices. India levies a duty of 0 to 7.5% on imports of medical devices, with most devices charged at 7.5%. Medical devices include consumables and disposables (gloves, syringes, PPP kits), electronic equipment, instruments, implants, and IVD reagents.














